Drug Catalog - Product Detail
TOPIRAMATE TAB USP 50 MG 1000CT
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 68462-0153-10 | GLENMARK PHARMACEUTICALS | 1000 | 50MG | TABLET |
PACKAGE FILES






Generic Name
TOPIRAMATE
Substance Name
TOPIRAMATE
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA077627
Description
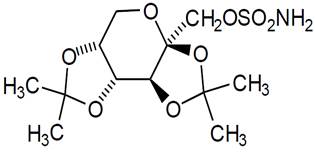
11 DESCRIPTION Topiramate, USP is a sulfamate-substituted monosaccharide. Topiramate Tablets, USP are available as 25 mg, 50 mg, 100 mg, and 200 mg round tablets for oral administration. Topiramate, USP is a white to off-white powder. It is freely soluble in dichloromethane. Topiramate, USP has the molecular formula C 12 H 21 NO 8 S and a molecular weight of 339.36. Topiramate, USP is designated chemically as 2,3:4,5-Di-O-isopropylidene-β-D-fructopyranose sulfamate and has the following structural formula: Topiramate tablets, USP contain the following inactive ingredients: hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, pregelatinized starch, polyethylene glycol, polysorbate 80, sodium starch glycolate, and titanium dioxide. The 50 mg tablets also contain FD&C yellow# 6 and iron oxide yellow for color. The 100 mg and 200 mg tablets also contain iron oxide red and iron oxide yellow for color. structure
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING 16.1 How Supplied Topiramate Tablets, USP are available as circular, biconvex, film-coated tablets in the following strengths and colors: 25 mg white tablets with ‘G’ engraved on one side and ‘25’ on the other side. Bottles of 60 count with desiccant (NDC 68462-108-60) Bottles of 500 count with desiccant (NDC 68462-108-05) Bottles of 1000 count with desiccant (NDC 68462-108-10) 50 mg yellow tablets with ‘G’ engraved on one side and ‘50’ on the other side. Bottles of 60 count with desiccant (NDC 68462-153-60) Bottles of 500 count with desiccant (NDC 68462-153-05) Bottles of 1000 count with desiccant (NDC 68462-153-10) 100 mg yellow tablets with ‘G’ engraved on one side and ‘100’ on the other side. Bottles of 60 count with desiccant (NDC 68462-109-60) Bottles of 500 count with desiccant (NDC 68462-109-05) Bottles of 1000 count with desiccant (NDC 68462-109-10) 200 mg pink tablets with ‘G’ engraved on one side and ‘200’ on the other side. Bottles of 60 count with desiccant (NDC 68462-110-60) Bottles of 500 count with desiccant (NDC 68462-110-05) Bottles of 1000 count with desiccant (NDC 68462-110-10) 16.2 Storage and Handling Topiramate tablets should be stored in tightly-closed containers at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
Indications & Usage
1 INDICATIONS AND USAGE Topiramate tablets are indicated for: • Epilepsy: initial monotherapy for the treatment of partial-onset or primary generalized tonic-clonic seizures in patients 2 years of age and older ( 1.1 ); adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, or seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older ( 1.2 ) • Preventive treatment of migraine in patients 12 years of age and older ( 1.3 ) 1.1 Monotherapy Epilepsy Topiramate tablets are indicated as initial monotherapy for the treatment of partial-onset or primary generalized tonic‑clonic seizures in patients 2 years of age and older. 1.2 Adjunctive Therapy Epilepsy Topiramate tablets are indicated as adjunctive therapy for the treatment of partial-onset seizures, primary generalized tonic-clonic seizures, and seizures associated with Lennox-Gastaut syndrome in patients 2 years of age and older. 1.3 Migraine Topiramate tablets are indicated for the preventive treatment of migraine in patients 12 years of age and older.
Dosage and Administration
2 DOSAGE AND ADMINISTRATION Topiramate tablets initial dose, titration, and recommended maintenance dose varies by indication and age group. See Full Prescribing Information for recommended dosage, and dosing considerations in patients with renal impairment, geriatric patients, and patients undergoing hemodialysis ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 , 2.6 ) 2.1 Dosing in Monotherapy Epilepsy Adults and Pediatric Patients 10 Years of Age and Older The recommended dose for topiramate tablets monotherapy in adults and pediatric patients 10 years of age and older is 400 mg/day in two divided doses. The dose should be achieved by titration according to the following schedule (Table 1): Table 1: Monotherapy Titration Schedule for Adults and Pediatric Patients 10 years and older Morning Dose Evening Dose Week 1 25 mg 25 mg Week 2 50 mg 50 mg Week 3 75 mg 75 mg Week 4 100 mg 100 mg Week 5 150 mg 150 mg Week 6 200 mg 200 mg Pediatric Patients 2 to 9 Years of Age Dosing in patients 2 to 9 years of age is based on weight. During the titration period, the initial dose of topiramate tablets is 25 mg/day nightly for the first week. Based upon tolerability, the dosage can be increased to 50 mg/day (25 mg twice daily) in the second week. Dosage can be increased by 25 to 50 mg/day each subsequent week as tolerated. Titration to the minimum maintenance dose should be attempted over 5 to 7 weeks of the total titration period. Based upon tolerability and clinical response, additional titration to a higher dose (up to the maximum maintenance dose) can be attempted at 25 to 50 mg/day weekly increments. The total daily dose should not exceed the maximum maintenance dose for each range of body weight (Table 2). Table 2: Monotherapy Target Total Daily Maintenance Dosing for Patients 2 to 9 Years of Age Weight (kg) Total Daily Dose (mg/day) * Minimum Maintenance Dose Total Daily Dose (mg/day) * Maximum Maintenance Dose Up to 11 150 250 12 to 22 200 300 23 to 31 200 350 32 to 38 250 350 Greater than 38 250 400 *Administered in two equally divided doses 2.2 Dosing in Adjunctive Therapy Epilepsy Adults (17 Years of Age and Older) The recommended total daily dose of topiramate tablets as adjunctive therapy in adults with partial-onset seizures or Lennox-Gastaut Syndrome is 200 to 400 mg/day in two divided doses, and 400 mg/day in two divided doses as adjunctive treatment in adults with primary generalized tonic-clonic seizures. Topiramate tablets should be initiated at 25 to 50 mg/day, followed by titration to an effective dose in increments of 25 to 50 mg/day every week. Titrating in increments of 25 mg/day every week may delay the time to reach an effective dose. Doses above 400 mg/day have not been shown to improve responses in adults with partial-onset seizures. Pediatric Patients 2 to 16 Years of Age The recommended total daily dose of topiramate tablets as adjunctive therapy for pediatric patients 2 to 16 years of age with partial-onset seizures, primary generalized tonic-clonic seizures, or seizures associated with Lennox-Gastaut syndrome is approximately 5 to 9 mg/kg/day in two divided doses. Titration should begin at 25 mg/day (or less, based on a range of 1 to 3 mg/kg/day) nightly for the first week. The dosage should then be increased at 1- or 2-week intervals by increments of 1 to 3 mg/kg/day (administered in two divided doses), to achieve optimal clinical response. Dose titration should be guided by clinical outcome. The total daily dose should not exceed 400 mg/day. 2.3 Dosing for the Preventive Treatment of Migraine The recommended total daily dose of topiramate tablets as treatment for patients 12 years of age and older for the preventive treatment of migraine is 100 mg/day administered in two divided doses (Table 3). The recommended titration rate for topiramate tablets for the preventive treatment of migraine is as follows: Table 3: Preventive Treatment of Migraine Titration Schedule for Patients 12 Years of Age and Older Morning Dose Evening Dose Week 1 None 25 mg Week 2 25 mg 25 mg Week 3 25 mg 50 mg Week 4 50 mg 50 mg Dose and titration rate should be guided by clinical outcome. If required, longer intervals between dose adjustments can be used. 2.4 Administration Information Topiramate tablets can be taken without regard to meals. Because of the bitter taste, tablets should not be broken. 2.5 Dosing in Patients with Renal Impairment In patients with renal impairment (creatinine clearance less than 70 mL/min/1.73 m 2 ), one-half of the usual adult dose of topiramate tablets are recommended [see Use in Specific Populations ( 8.5 , 8.6 ), Clinical Pharmacology ( 12.3 )] . 2.6 Dosing in Patients Undergoing Hemodialysis To avoid rapid drops in topiramate plasma concentration during hemodialysis, a supplemental dose of topiramate tablets may be required. The actual adjustment should take into account 1) the duration of dialysis period, 2) the clearance rate of the dialysis system being used, and 3) the effective renal clearance of topiramate in the patient being dialyzed [see Use in Specific Populations ( 8.7 ), Clinical Pharmacology ( 12.3 )] .
