Drug Catalog - Product Detail
PREZCOBIX 150-800MG TAB 30CT
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 59676-0575-30 | JANSSEN PRODUCTS | 30 | 800-150MG | TABLET |
PACKAGE FILES









Generic Name
DARUNAVIR ETHANOLATE AND COBICISTAT
Substance Name
COBICISTAT
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
NDA205395
Description
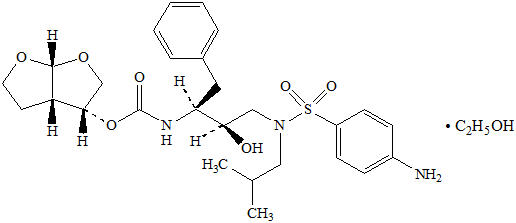
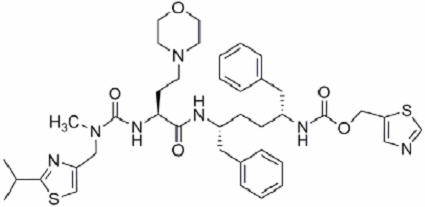
11 DESCRIPTION PREZCOBIX and PREZCOBIX PED are fixed-dose combination products containing darunavir and cobicistat. Darunavir is an inhibitor of the human immunodeficiency virus (HIV-1) protease. Cobicistat is a mechanism-based inhibitor of cytochrome P450 (CYP) enzymes of the CYP3A family. Darunavir : Darunavir, in the form of darunavir ethanolate, has the following chemical name: [(1 S ,2 R )-3-[[(4-aminophenyl)sulfonyl](2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]-carbamic acid (3 R ,3a S ,6a R )-hexahydrofuro[2,3- b ]furan-3-yl ester monoethanolate. Its molecular formula is C 27 H 37 N 3 O 7 S ∙ C 2 H 5 OH and its molecular weight is 593.73. Darunavir ethanolate has the following structural formula: Cobicistat : Cobicistat is adsorbed onto silicon dioxide. The chemical name for cobicistat is 1,3-thiazol-5-ylmethyl[(2 R ,5 R )-5-{[(2 S )2-[(methyl{[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl}carbamoyl)amino]-4-(morpholin-4yl)butanoyl]amino}-1,6-diphenylhexan-2-yl]carbamate. It has a molecular formula of C 40 H 53 N 7 O 5 S 2 and a molecular weight of 776.0. It has the following structural formula: PREZCOBIX ® 800 mg darunavir/150 mg cobicistat tablets are for oral administration. Each tablet contains darunavir ethanolate equivalent to 800 mg of darunavir and 150 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, crospovidone, hypromellose, magnesium stearate, and silicified microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. PREZCOBIX ® 675 mg darunavir/150 mg cobicistat tablets are for oral administration. Each tablet contains darunavir ethanolate equivalent to 675 mg of darunavir and 150 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide yellow, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. PREZCOBIX ® PED 600 mg darunavir/90 mg cobicistat tablets for oral suspension are for oral administration. Each tablet for oral suspension contains darunavir ethanolate equivalent to 600 mg of darunavir and 90 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, silicified microcrystalline cellulose, strawberry flavor, and sucralose. The tablets are film-coated with a coating material containing glycerol monocaprylocaprate type 1, iron oxide red, iron oxide black, macrogol polyvinyl alcohol graft copolymer, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. Chemical Structure Chemical Structure
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING PREZCOBIX Tablets PREZCOBIX ® (darunavir and cobicistat) tablets, 800/150 mg, are supplied as pink, oval-shaped, film-coated tablets debossed with "800" on one side and "TG" on the other side. Bottle of 30 tablets with child-resistant closure (NDC 59676-575-30). PREZCOBIX ® (darunavir and cobicistat) tablets, 675/150 mg, are supplied as green to dark green, oval-shaped, scored film-coated tablet debossed with "675" on one side and "TG" on the other side. Bottle of 30 tablets with child-resistant closure (NDC 59676-578-30). Storage of PREZCOBIX tablets: Store at 20 °C to 25 °C (between 68 °F to 77 °F); with excursions permitted to 15 °C to 30 °C (59 °F to 86 °F) [see USP Controlled Room Temperature]. PREZCOBIX PED Tablets for Oral Suspension PREZCOBIX ® PED (darunavir and cobicistat) tablets for oral suspension, 600/90 mg, are supplied as reddish purple, oval-shaped, film-coated tablets debossed with "690" on one side and "TG" on the other side. Bottle of 30 tablets with child-resistant closure (NDC 59676-577-30). Storage of PREZCOBIX PED tablets for oral suspension: Store in the original container in order to protect from light. Keep the container tightly closed. Store at 20 °C to 25 °C (between 68 °F to 77 °F); with excursions permitted to 15 °C to 30 °C (59 °F to 86 °F) [see USP Controlled Room Temperature]. Keep PREZCOBIX, PREZCOBIX PED, and all medicines out of reach of children.
Indications & Usage
1 INDICATIONS AND USAGE PREZCOBIX and PREZCOBIX PED are indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) in treatment-naïve and treatment-experienced adults and pediatric patients 3 years of age and older weighing at least 15 kg with no darunavir resistance-associated substitutions (V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V) [see Use in Specific Populations (8.4) and Clinical Studies (14) ] . PREZCOBIX and PREZCOBIX PED are a two-drug combination of darunavir, a human immunodeficiency virus (HIV-1) protease inhibitor, and cobicistat, a CYP3A inhibitor, and are indicated for the treatment of HIV-1 in treatment-naïve and treatment-experienced adults and pediatric patients 3 years of age and older weighing at least 15 kg with no darunavir resistance-associated substitutions (V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V). ( 1 )
Dosage and Administration
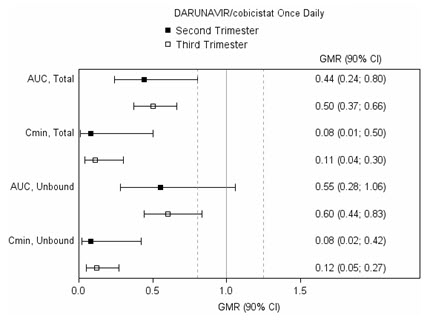
2 DOSAGE AND ADMINISTRATION Recommended dosage: Adults and pediatric patients weighing at least 40 kg: One 800 mg/150 mg tablet taken once daily with food. ( 2.1 , 2.2 ) Pediatric patients weighing at least 25 kg to less than 40 kg: One 675 mg/150 mg tablet taken once daily with food. ( 2.1 , 2.2 ) Pediatric patients 3 years of age and older weighing at least 15 kg to less than 25 kg: One 600 mg/90 mg tablet for oral suspension taken once daily with food. PREZCOBIX PED must be dispersed in drinking water and taken immediately with food. ( 2.2 , 2.3 ) Testing Prior to Initiation: HIV genotypic testing is recommended for antiretroviral treatment experienced patients. Assess estimated creatinine clearance in all patients prior to starting PREZCOBIX or PREZCOBIX PED. When used with tenofovir disoproxil fumarate (TDF): Assess urine glucose and urine protein at baseline and monitor creatinine clearance, urine glucose, and urine protein. Monitor serum phosphorus in patients with or at risk for renal impairment. ( 2.5 ) 2.1 Overview of Different Dosage Forms Two different dosage forms are available: PREZCOBIX Tablets: 800 mg/150 mg film-coated tablets for adults and pediatric patients weighing at least 40 kg. 675 mg/150 mg film-coated tablets for pediatric patients weighing at least 25 kg to less than 40 kg. PREZCOBIX PED Tablets for Oral Suspension: 600 mg/90 mg film-coated tablet for oral suspension for pediatric patients aged 3 years and older weighing at least 15 kg to less than 25 kg [see Dosage and Administration (2.2 , 2.3) ] . 2.2 Recommended Dosage in Adults and Pediatrics 3 Years of Age and Older Weighing at Least 15 kg The recommended dosages for adults and pediatric patients weighing at least 15 kg are shown in Table 1. The pediatric dose is based on weight. PREZCOBIX and PREZCOBIX PED are taken orally with food once daily in conjunction with other antiretroviral agents. Table 1: Recommended Dosages of PREZCOBIX and PREZCOBIX PED in Adults and Pediatric Patients Weighing at Least 15 kg, who are Treatment-Naïve or Treatment-Experienced Without DRV RAMs DRV-resistance-associated mutations (RAMs): V11I, V32I, L33F, I47V, I50V, I54M, I54L, T74P, L76V, I84V, L89V. Patient Population Total Daily Dose Adults and pediatric patients weighing at least 40 kg One PREZCOBIX 800 mg darunavir/150 mg cobicistat tablet Pediatric Patients weighing at least 25 kg to less than 40 kg One PREZCOBIX 675 mg darunavir/150 mg cobicistat tablet Pediatric Patients aged 3 years and older weighing at least 15 kg to less than 25 kg One PREZCOBIX PED 600 mg darunavir/90 mg cobicistat tablet for oral suspension Before prescribing PREZCOBIX 675 mg/150 mg tablets, children should be assessed for the ability to swallow tablets. For patients unable to swallow the 675 mg/150 mg tablet whole, the scored tablet may be split by hand into two pieces. Each piece should be consumed immediately after splitting to ensure the entire dose is administered. The score line is only to facilitate breaking for ease of swallowing and not to divide into equal doses. 2.3 Preparation and Administration Instructions for PREZCOBIX PED Advise patients or caregivers of patients taking PREZCOBIX PED to refer to the Instructions for Use to properly prepare and take the medication. PREZCOBIX PED must be dispersed in drinking water and taken immediately as described below. If not taken immediately, then the oral suspension should be discarded, and a new dose of medicine should be prepared. The patient should not crush, chew or swallow the PREZCOBIX PED tablet for oral suspension. The following instructions should be followed: Place the tablet for oral suspension in a cup, add 30 mL (2 tablespoons) of non-carbonated room temperature drinking water. Stir well with a spoon until the tablet is completely dispersed. The prepared medicine will appear reddish purple. Take all the prepared medicine immediately or to aid in administration, the prepared medicine can be further diluted with 10 mL (2 teaspoons) of water, orange juice, or 1 teaspoon of applesauce or yogurt. Mix and take the entire mixture immediately. Add another 5 mL (1 teaspoon) of non-carbonated drinking water to the cup, swirl and drink completely. This step may be repeated as needed to ensure the entire dose is consumed. 2.4 Testing Prior to Initiation of PREZCOBIX or PREZCOBIX PED HIV Genotypic Testing HIV genotypic testing is recommended for antiretroviral treatment-experienced patients. However, when HIV genotypic testing is not feasible, PREZCOBIX or PREZCOBIX PED can be used in protease inhibitor-naïve patients, but is not recommended in protease inhibitor-experienced patients. Creatinine Clearance Prior to starting PREZCOBIX or PREZCOBIX PED, assess estimated creatinine clearance because cobicistat decreases estimated creatinine clearance due to inhibition of tubular secretion of creatinine without affecting actual renal glomerular function [see Warnings and Precautions (5.3) ] . When co-administering PREZCOBIX or PREZCOBIX PED with tenofovir disoproxil fumarate (tenofovir DF) assess estimated creatinine clearance, urine glucose, and urine protein at baseline [see Warnings and Precautions (5.4) ] . 2.5 Not Recommended in Severe Renal Impairment PREZCOBIX or PREZCOBIX PED co-administered with tenofovir DF is not recommended in patients who have an estimated creatinine clearance below 70 mL per minute [see Warnings and Precautions (5.4) and Adverse Reactions (6) ] . 2.6 Not Recommended in Severe Hepatic Impairment PREZCOBIX or PREZCOBIX PED is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3) ] . 2.7 Not Recommended During Pregnancy PREZCOBIX or PREZCOBIX PED is not recommended during pregnancy because of substantially lower exposures of darunavir and cobicistat during the second and third trimesters [see Use in Specific Populations (8.1) and Clinical Pharmacology (12.3) ] . PREZCOBIX or PREZCOBIX PED should not be initiated in pregnant individuals. An alternative regimen is recommended for those who become pregnant during therapy with PREZCOBIX or PREZCOBIX PED.
