Drug Catalog - Product Detail
NESINA TABS 25MG 30CT
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 64764-0250-30 | TAKEDA PHARMACEUTICALS | 30 | 25MG | TABLET |
PACKAGE FILES






Generic Name
ALOGLIPTIN
Substance Name
ALOGLIPTIN BENZOATE
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
NDA022271
Description
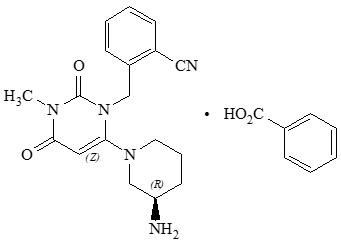
11 DESCRIPTION NESINA tablets contain the active ingredient alogliptin, which is a selective, orally bioavailable inhibitor of the enzymatic activity of DPP-4. Chemically, alogliptin is prepared as a benzoate salt, which is identified as 2-({6-[(3 R )-3-aminopiperidin-1-yl]-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2 H )-yl}methyl)benzonitrile monobenzoate. It has a molecular formula of C 18 H 21 N 5 O 2 ∙C 7 H 6 O 2 and a molecular weight of 461.51 daltons. The structural formula is: Alogliptin benzoate is a white to off-white crystalline powder containing one asymmetric carbon in the aminopiperidine moiety. It is soluble in dimethylsulfoxide, sparingly soluble in water and methanol, slightly soluble in ethanol and very slightly soluble in octanol and isopropyl acetate. Each NESINA tablet contains 34 mg, 17 mg or 8.5 mg alogliptin benzoate, which is equivalent to 25 mg, 12.5 mg or 6.25 mg, respectively, of alogliptin and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, mannitol, and microcrystalline cellulose. In addition, the film coating contains the following inactive ingredients: ferric oxide (red or yellow), hypromellose, polyethylene glycol, and titanium dioxide and is marked with printing ink (Gray F1). Chemical Structure
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING NESINA tablets are available as film-coated tablets containing 25 mg, 12.5 mg or 6.25 mg of alogliptin as follows: 25 mg tablet: light red, oval, biconvex, film-coated, with "TAK ALG-25" printed on one side, available in: NDC 64764-250-30 Bottles of 30 tablets NDC 64764-250-90 Bottles of 90 tablets NDC 64764-250-50 Bottles of 500 tablets 12.5 mg tablet: yellow, oval, biconvex, film-coated, with "TAK ALG-12.5" printed on one side, available in: NDC 64764-125-30 Bottles of 30 tablets NDC 64764-125-90 Bottles of 90 tablets NDC 64764-125-50 Bottles of 500 tablets 6.25 mg tablet: light pink, oval, biconvex, film-coated, with "TAK ALG-6.25" printed on one side, available in: NDC 64764-625-30 Bottles of 30 tablets NDC 64764-625-90 Bottles of 90 tablets Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Indications & Usage
1 INDICATIONS AND USAGE NESINA ® is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. NESINA is a dipeptidyl peptidase-4 (DPP-4) inhibitor indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. ( 1 ) Limitations of Use: Should not be used in patients with type 1 diabetes mellitus. ( 1 ) Limitations of Use NESINA is not recommended for use in patients with type 1 diabetes mellitus.
Dosage and Administration
2 DOSAGE AND ADMINISTRATION The recommended dosage in patients with normal renal function or mild renal impairment is 25 mg orally once daily. ( 2.1 ) Can be taken with or without food. ( 2.1 ) Adjust dosage if moderate or severe renal impairment or end-stage renal disease (ESRD). ( 2.2 ) Degree of Renal Impairment Creatinine Clearance (mL/min) Recommended Dosage Moderate ≥30 to <60 12.5 mg once daily Severe/ESRD <30 6.25 mg once daily 2.1 Recommended Dosage The recommended dosage of NESINA is 25 mg taken orally once daily. Do not split tablets. NESINA may be taken with or without food [see Clinical Pharmacology (12.3) ] . Instruct patients if a dose is missed, not to double their next dose. 2.2 Patients with Renal Impairment Assess renal function prior to initiation of NESINA and periodically thereafter [see Use in Specific Populations (8.6) ] . No dose adjustment of NESINA is necessary for patients with mild renal impairment (creatinine clearance [CrCl] ≥60 mL/min). The dose of NESINA is 12.5 mg once daily for patients with moderate renal impairment (CrCl ≥30 to <60 mL/min). The dose of NESINA is 6.25 mg once daily for patients with severe renal impairment (CrCl ≥15 to <30 mL/min) or with end-stage renal disease (ESRD) (CrCl <15 mL/min or requiring hemodialysis). NESINA may be administered without regard to the timing of dialysis. NESINA has not been studied in patients undergoing peritoneal dialysis [see Use in Specific Populations (8.6) , Clinical Pharmacology (12.3) ] .
