Drug Catalog - Product Detail
EZETIMIBE 10MG TB 500CT
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 00591-3713-05 | ACTAVIS PHARMA | 500 | 10MG | TABLET |
PACKAGE FILES

Generic Name
EZETIMIBE
Substance Name
EZETIMIBE
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA200831
Description
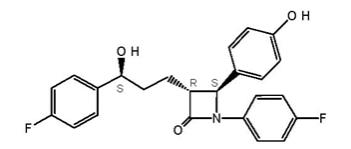
11 DESCRIPTION Ezetimibe, USP is a dietary cholesterol absorption inhibitor. The chemical name of ezetimibe is 1-(4-fluorophenyl)-3(R)-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4(S)-(4-hydroxyphenyl)-2-azetidinone. The empirical formula is C 24 H 21 F 2 NO 3 . Its molecular weight is 409.4 and its structural formula is: Ezetimibe, USP is a white, crystalline powder that is freely to very soluble in ethanol, methanol, and acetone and practically insoluble in water. Ezetimibe, USP has a melting point of about 163°C and is stable at ambient temperature. Ezetimibe, USP is available as a tablet for oral administration containing 10 mg of ezetimibe and the following inactive ingredients: croscarmellose sodium, crospovidone, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. structural formula
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING Ezetimibe tablets, USP 10 mg, are white to off white, capsule-shaped tablets debossed with " 713 " on one side and plain on the other side. They are supplied as follows: NDC 0591-3713-30 bottles of 30 NDC 0591-3713-19 bottles of 90 NDC 0591-3713-05 bottles of 500 Storage Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
Indications & Usage
1 INDICATIONS AND USAGE Ezetimibe tablets are indicated: In combination with a statin, or alone when additional low-density lipoprotein cholesterol (LDL-C) lowering therapy is not possible, as an adjunct to diet to reduce elevated LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH). In combination with a statin as an adjunct to diet to reduce elevated LDL-C in pediatric patients 10 years of age and older with HeFH. In combination with fenofibrate as an adjunct to diet to reduce elevated LDL-C in adults with mixed hyperlipidemia. In combination with a statin, and other LDL-C lowering therapies, to reduce elevated LDL-C levels in adults and in pediatric patients 10 years of age and older with homozygous familial hypercholesterolemia (HoFH). As an adjunct to diet for the reduction of elevated sitosterol and campesterol levels in adults and in pediatric patients 9 years of age and older with homozygous familial sitosterolemia. When ezetimibe tablets are used in combination with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for information on the safe and effective use. Ezetimibe is indicated ( 1 ): In combination with a statin, or alone when additional low density lipoprotein cholesterol (LDL-C) lowering therapy is not possible, as an adjunct to diet to reduce elevated LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH). In combination with a statin as an adjunct to diet to reduce elevated LDL-C in pediatric patients 10 years of age and older with HeFH. In combination with fenofibrate as an adjunct to diet to reduce elevated LDL-C in adults with mixed hyperlipidemia. In combination with a statin, and other LDL-C lowering therapies, to reduce elevated LDL-C levels in adults and in pediatric patients 10 years of age and older with homozygous familial hypercholesterolemia (HoFH). As an adjunct to diet for the reduction of elevated sitosterol and campesterol levels in adults and in pediatric patients 9 years of age and older with homozygous familial sitosterolemia. When ezetimibe is used in combination with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for information on the safe and effective use ( 1 ).
Dosage and Administration
2 DOSAGE AND ADMINISTRATION The recommended dose of ezetimibe tablets is 10 mg orally once daily, administered with or without food. If as dose is missed, take the missed dose as soon as possible. Do not double the next dose. Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating ezetimibe tablets. Administer ezetimibe tablets at least 2 hours before or 4 hours after administration of a bile acid sequestrant [see Drug Interactions ( 7 )] . 10-mg orally once daily, with or without food ( 2 ) Administer ezetimibe tablets either ≥2 hours before or ≥4 hours after administration of a bile acid sequestrant. ( 2 ) Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating ezetimibe tablets. ( 2 )
