Drug Catalog - Product Detail
DOXEPIN HCL CREAM 5% 45GM
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 00378-8117-45 | MYLAN | 45 | 5% | CREAM |
PACKAGE FILES
Generic Name
DOXEPIN HYDROCHLORIDE
Substance Name
DOXEPIN HYDROCHLORIDE
Product Type
HUMAN PRESCRIPTION DRUG
Route
TOPICAL
Application Number
NDA020126
Description
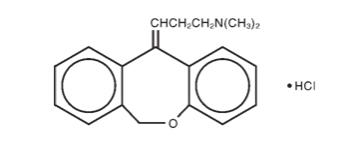
DESCRIPTION Doxepin Hydrochloride Cream, 5% is a topical cream. Each gram contains: 50 mg of doxepin hydrochloride (equivalent to 44.3 mg of doxepin). Doxepin hydrochloride, USP is one of a class of agents known as dibenzoxepin tricyclic antidepressant compounds. It is an isomeric mixture of N,N-dimethyldibenz[ b,e ]oxepin-Δ11(6H),γ-propylamine hydrochloride. Doxepin hydrochloride has a molecular formula of C 19 H 21 NO•HCl and a molecular weight of 316. Doxepin Hydrochloride Cream, 5% also contains sorbitol, cetyl alcohol, isopropyl myristate, glyceryl stearate, PEG-100 stearate, petrolatum, benzyl alcohol, titanium dioxide and purified water. Doxepin hydrochloride is one of a class of agents known as dibenzoxepin tricyclic antidepressant compounds. It is an isomeric mixture of N,N-dimethyldibenz[b,e]oxepin-Δ11(6H),γ-propylamine hydrochloride. Doxepin hydrochloride has an empirical formula of C19H21NO•HCl and a molecular weight of 316.
How Supplied
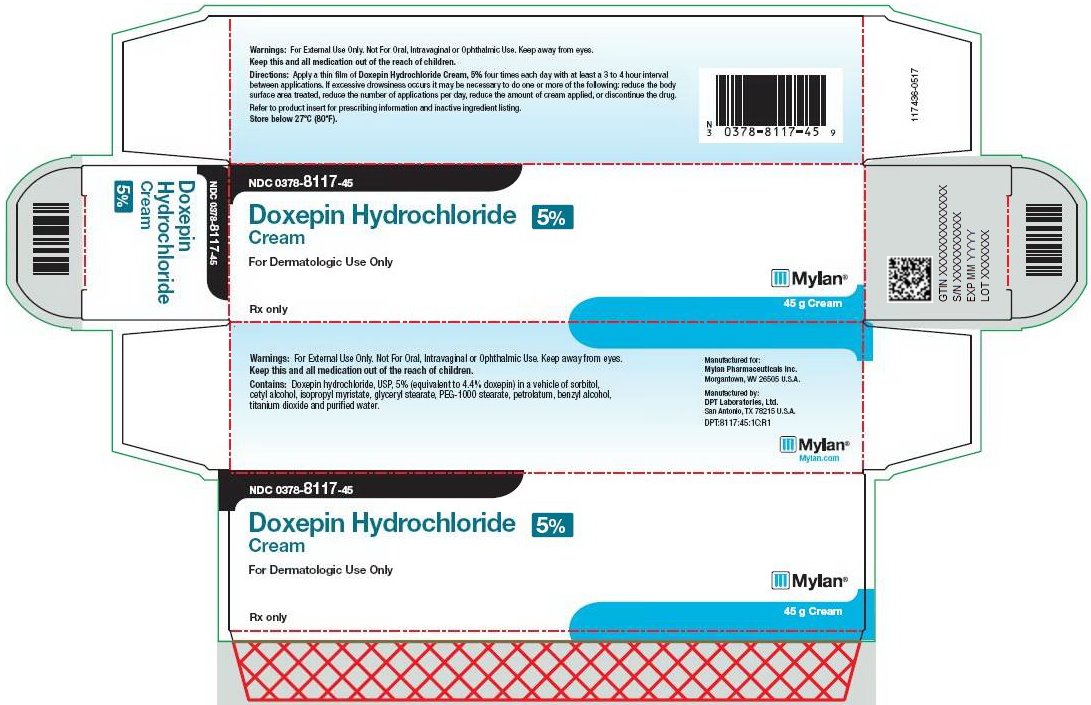
HOW SUPPLIED Each gram of Doxepin Hydrochloride Cream, 5% contains 50 mg of doxepin hydrochloride, USP equivalent to 44.3 mg of doxepin. Doxepin Hydrochloride Cream, 5% is a soft white cream available as follows: NDC 0378-8117-45 carton containing one 45 g tube Store below 27ºC (80ºF). Manufactured for: Mylan Pharmaceuticals Inc. Morgantown, WV 26505 U.S.A. Manufactured by: DPT Laboratories, Ltd. San Antonio, TX 78215 140902-0517 Revised: 5/2017 DPT:DOXCR:R1
Indications & Usage
INDICATIONS AND USAGE Doxepin Hydrochloride Cream, 5% is indicated for the short-term (up to 8 days) management of moderate pruritus in adult patients with atopic dermatitis or lichen simplex chronicus. (See DOSAGE AND ADMINISTRATION .)
Dosage and Administration
DOSAGE AND ADMINISTRATION A thin film of Doxepin Hydrochloride Cream, 5% should be applied four times each day with at least a 3 to 4 hour interval between applications. There are no data to establish the safety and effectiveness of Doxepin Hydrochloride Cream, 5% when used for greater than 8 days. Chronic use beyond eight days may result in higher systemic levels and should be avoided. Use of Doxepin Hydrochloride Cream, 5% for longer than 8 days may result in an increased likelihood of contact sensitization. The risk for sedation may increase with greater body surface area application of Doxepin Hydrochloride Cream, 5% (See WARNINGS section ). Clinical experience has shown that drowsiness is significantly more common in patients applying Doxepin Hydrochloride Cream, 5% to over 10% of body surface area; therefore, patients with greater than 10% of body surface area (see WARNINGS section ) affected should be particularly cautioned concerning possible drowsiness and other systemic adverse effects of doxepin. If excessive drowsiness occurs, it may be necessary to do one or more of the following: reduce the body surface area treated, reduce the number of applications per day, reduce the amount of cream applied, or discontinue the drug. Occlusive dressings may increase the absorption of most topical drugs; therefore, occlusive dressings should not be utilized with Doxepin Hydrochloride Cream, 5%.
