Drug Catalog - Product Detail
CLINDAMYCIN PALMITATE HCL FOR ORAL SOLUTIONUSP 75MG/5ML SOL 100ML
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 64980-0511-10 | RISING PHARMACEUTICALS | 100 | 75MG/5ML | SOLUTION |
PACKAGE FILES

Generic Name
CLINDAMYCIN PALMITATE HYDROCHLORIDE
Substance Name
CLINDAMYCIN PALMITATE HYDROCHLORIDE
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA201821
Description
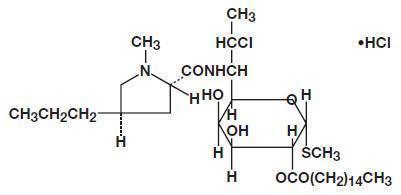
DESCRIPTION Clindamycin palmitate hydrochloride is a water soluble hydrochloride salt of the ester of clindamycin and palmitic acid. Clindamycin is a semisynthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent compound lincomycin. The structural formula is represented below: The chemical name for clindamycin palmitate hydrochloride is Methyl 7-chloro-6, 7, 8-trideoxy-6-(1-methyl- trans -4-propyl-L-2-pyrrolidinecarboxamido)-1-thio-L- threo -α-D- galacto -octopyranoside 2-palmitate monohydrochloride. Clindamycin palmitate hydrochloride for oral solution (Pediatric) flavored granules contain clindamycin palmitate hydrochloride for reconstitution. Each 5 mL contains the equivalent of 75 mg clindamycin. Inactive ingredients: natural cherry flavor, dextrin, ethylparaben, poloxamer 188, simethicone, sucrose. structure
How Supplied
HOW SUPPLIED Clindamycin palmitate hydrochloride for oral solution, (Pediatric) flavored granules is available in bottles of 100 mL (NDC 64980-511-10). When reconstituted as directed, each bottle yields a solution containing 75 mg of clindamycin per 5 mL. Rx only
Indications & Usage
INDICATIONS AND USAGE Clindamycin palmitate hydrochloride for oral solution, USP (Pediatric) (clindamycin palmitate HCl) is indicated in the treatment of serious infections caused by susceptible anaerobic bacteria. Clindamycin is also indicated in the treatment of serious infections due to susceptible strains of streptococci, pneumococci and staphylococci. Its use should be reserved for penicillin-allergic patients or other patients for whom, in the judgment of the physician, a penicillin is inappropriate. Because of the risk of colitis, as described in the BOXED WARNING , before selecting clindamycin the physician should consider the nature of the infection and the suitability of less toxic alternatives (e.g., erythromycin). Anaerobes: Serious respiratory tract infections such as empyema, anaerobic pneumonitis and lung abscess; serious skin and soft tissue infections; septicemia; intra-abdominal infections such as peritonitis and intra-abdominal abscess (typically resulting from anaerobic organisms resident in the normal gastrointestinal tract); infections of the female pelvis and genital tract such as endometritis, nongonococcal tubo-ovarian abscess, pelvic cellulitis and postsurgical vaginal cuff infection. Streptococci: Serious respiratory tract infections; serious skin and soft tissue infections. Staphylococci: Serious respiratory tract infections; serious skin and soft tissue infections. Pneumococci: Serious respiratory tract infections. Bacteriologic studies should be performed to determine the causative organisms and their susceptibility to clindamycin. To reduce the development of drug-resistant bacteria and maintain the effectiveness of Clindamycin palmitate hydrochloride for oral solution (Pediatric) and other antibacterial drugs, clindamycin palmitate hydrochloride for oral solution (Pediatric) should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Dosage and Administration
DOSAGE AND ADMINISTRATION If significant diarrhea occurs during therapy, this antibiotic should be discontinued (see BOXED WARNING ). Concomitant administration of food does not adversely affect the absorption of clindamycin palmitate HCl contained in clindamycin palmitate hydrochloride for oral solution (Pediatric) flavored granules. Serious infections: 8-12 mg/kg/day (4-6 mg/lb/day) divided into 3 or 4 equal doses. Severe infections: 13-16 mg/kg/day (6.5-8 mg/lb/day) divided into 3 or 4 equal doses. More severe infections: 17-25 mg/kg/day (8.5-12.5 mg/lb/day) divided into 3 or 4 equal doses. In pediatric patients weighing 10 kg or less, ½ teaspoon (37.5 mg) three times a day should be considered the minimum recommended dose. Clindamycin should be dosed based on total body weight regardless of obesity. Serious infections due to anaerobic bacteria are usually treated with clindamycin injection. However, in clinically appropriate circumstances, the physician may elect to initiate treatment or continue treatment with clindamycin palmitate hydrochloride for oral solution (Pediatric). NOTE: In cases of β-hemolytic streptococcal infections, treatment should be continued for at least 10 days. Reconstitution Instructions: When reconstituted with water as follows, each 5 mL (teaspoon) of solution contains clindamycin palmitate HCl equivalent to 75 mg clindamycin. Reconstitute bottles of 100 mL with 75 mL of water. Add a large portion of the water and shake vigorously; add the remainder of the water and shake until the solution is uniform. Storage Conditions: Store at controlled room temperature 20° to 25°C (68° to 77°F) [see USP]. Do NOT refrigerate the reconstituted solution; when chilled, the solution may thicken and be difficult to pour. The solution is stable for 2 weeks at room temperature.
