Drug Catalog - Product Detail
CEFUROXIME AXETIL TB 500MG 60
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 65862-0700-60 | AUROBINDO PHARMA | 60 | 500MG | TABLET |
PACKAGE FILES



Generic Name
Substance Name
Product Type
Route
Application Number
Description
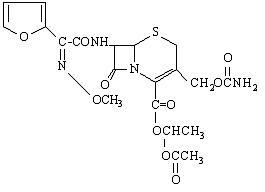
11 DESCRIPTION Cefuroxime axetil tablets, USP contain cefuroxime as cefuroxime axetil. Cefuroxime axetil is a semisynthetic, cephalosporin antibacterial drug for oral administration. The chemical name of cefuroxime axetil (1-(acetyloxy) ethyl ester of cefuroxime) is ( RS )-1-hydroxyethyl (6 R ,7 R )-7-[2-(2-furyl)glyoxyl-amido]-3-(hydroxymethyl)-8-oxo-5-thia-1-azabicyclo[4.2.0]-oct-2-ene-2-carboxylate, 7 2 -( Z )-( O -methyl-oxime), 1-acetate 3-carbamate. Its molecular formula is C 20 H 22 N 4 O 10 S, and it has a molecular weight of 510.48. Cefuroxime axetil is in the amorphous form and has the following structural formula: Tablets are film-coated and contain the equivalent of 125 mg, 250 mg or 500 mg of cefuroxime as cefuroxime axetil USP. Tablets contain the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, hydrogenated vegetable oil, hypromellose, microcrystalline cellulose, polyethylene glycol, sodium lauryl sulfate, and titanium dioxide. Chemical Structure
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING Cefuroxime Axetil Tablets USP, 125 mg of cefuroxime (as cefuroxime axetil), are white to off-white, film-coated, capsule shaped tablets with ‘A32’ debossed on one side and plain on the other side. 20 Tablets/Bottle NDC 65862-698-20 60 Tablets/Bottle NDC 65862-698-60 Cefuroxime Axetil Tablets USP, 250 mg of cefuroxime (as cefuroxime axetil), are white to off-white, film-coated, capsule shaped tablets with ‘A33’ debossed on one side and plain on the other side. 20 Tablets/Bottle NDC 65862-699-20 60 Tablets/Bottle NDC 65862-699-60 Cefuroxime Axetil Tablets USP, 500 mg of cefuroxime (as cefuroxime axetil), are white to off-white, film-coated, capsule shaped tablets with ‘A34’ debossed on one side and plain on the other side. 20 Tablets/Bottle NDC 65862-700-20 60 Tablets/Bottle NDC 65862-700-60 Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Replace cap securely after each opening.
Indications & Usage
1 INDICATIONS AND USAGE Cefuroxime axetil tablets are a cephalosporin antibacterial drug indicated for the treatment of the following infections due to susceptible bacteria: ( 1 ) Pharyngitis/tonsillitis (adults and pediatric patients) ( 1.1 ) Acute bacterial otitis media (pediatric patients) ( 1.2 ) Acute bacterial maxillary sinusitis (adults and pediatric patients) ( 1.3 ) Acute bacterial exacerbations of chronic bronchitis (adults and pediatric patients 13 years and older) ( 1.4 ) Uncomplicated skin and skin-structure infections (adults and pediatric patients 13 years and older) ( 1.5 ) Uncomplicated urinary tract infections (adults and pediatric patients 13 years and older) ( 1.6 ) Uncomplicated gonorrhea (adults and pediatric patients 13 years and older) ( 1.7 ) Early Lyme disease (adults and pediatric patients 13 years and older) ( 1.8 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefuroxime axetil tablets and other antibacterial drugs, cefuroxime axetil tablets should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. 1.1 Pharyngitis/Tonsillitis Cefuroxime axetil tablets are indicated for the treatment of adult patients and pediatric patients (13 years and older) with mild-to-moderate pharyngitis/tonsillitis caused by susceptible strains of Streptococcus pyogenes . Limitations of Use The efficacy of cefuroxime axetil tablets in the prevention of rheumatic fever was not established in clinical trials. The efficacy of cefuroxime axetil tablets in the treatment of penicillin-resistant strains of Streptococcus pyogenes has not been demonstrated in clinical trials. 1.2 Acute Bacterial Otitis Media Cefuroxime axetil tablets are indicated for the treatment of pediatric patients (who can swallow tablets whole) with acute bacterial otitis media caused by susceptible strains of Streptococcus pneumoniae, Haemophilus influenzae (including β-lactamase–producing strains), Moraxella catarrhalis (including β-lactamase–producing strains), or Streptococcus pyogenes . 1.3 Acute Bacterial Maxillary Sinusitis Cefuroxime axetil tablets are indicated for the treatment of adult and pediatric patients (13 years and older) with mild-to-moderate acute bacterial maxillary sinusitis caused by susceptible strains of Streptococcus pneumoniae or Haemophilus influenzae (non - β-lactamase–producing strains only). Limitations of Use The effectiveness of cefuroxime axetil tablets for sinus infections caused by β-lactamase–producing Haemophilus influenzae or Moraxella catarrhalis in patients with acute bacterial maxillary sinusitis was not established due to insufficient numbers of these isolates in the clinical trials [see Clinical Studies (14.1) ] . 1.4 Acute Bacterial Exacerbations of Chronic Bronchitis Cefuroxime axetil tablets are indicated for the treatment of adult patients and pediatric patients (aged 13 and older) with mild-to-moderate acute bacterial exacerbations of chronic bronchitis caused by susceptible strains of Streptococcus pneumoniae , Haemophilus influenzae (β-lactamase–negative strains), or Haemophilus parainfluenzae (β-lactamase–negative strains). 1.5 Uncomplicated Skin and Skin-Structure Infections Cefuroxime axetil tablets are indicated for the treatment of adult patients and pediatric patients (aged 13 and older) with uncomplicated skin and skin-structure infections caused by susceptible strains of Staphylococcus aureus (including β - lactamase–producing strains) or Streptococcus pyogenes. 1.6 Uncomplicated Urinary Tract Infections Cefuroxime axetil tablets are indicated for the treatment of adult patients and pediatric patients (aged 13 and older) with uncomplicated urinary tract infections caused by susceptible strains of Escherichia coli or Klebsiella pneumoniae . 1.7 Uncomplicated Gonorrhea Cefuroxime axetil tablets are indicated for the treatment of adult patients and pediatric patients (aged 13 and older) with uncomplicated gonorrhea, urethral and endocervical, caused by penicillinase-producing and non-penicillinase–producing susceptible strains of Neisseria gonorrhoeae and uncomplicated gonorrhea, rectal, in females, caused by non-penicillinase–producing susceptible strains of Neisseria gonorrhoeae . 1.8 Early Lyme Disease (erythema migrans) Cefuroxime axetil tablets are indicated for the treatment of adult patients and pediatric patients (aged 13 and older) with early Lyme disease (erythema migrans) caused by susceptible strains of Borrelia burgdorferi. 1.10 Usage To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefuroxime axetil tablets and other antibacterial drugs, cefuroxime axetil tablets should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Dosage and Administration
2 DOSAGE AND ADMINISTRATION Tablets and oral suspension are not bioequivalent and are therefore not substitutable on a milligram-per-milligram basis. ( 2.1 ) Administer tablets with or without food. ( 2.2 ) Administer cefuroxime axetil tablets as described in the dosage guidelines. ( 2.2 ) Dosage adjustment is required for patients with impaired renal function. ( 2.5 ) Adult Patients and Pediatric Patients Dosage Guidelines for Cefuroxime Axetil Tablets Infection Dosage Duration (Days) Adults and Adolescents (13 years and older) Pharyngitis/tonsillitis (mild to moderate) 250 mg every 12 hours 10 Acute bacterial maxillary sinusitis (mild to moderate) 250 mg every 12 hours 10 Acute bacterial exacerbations of chronic bronchitis (mild to moderate) 250 mg or 500 mg every 12 hours 10 Uncomplicated skin and skin-structure infections 250 mg or 500 mg every 12 hours 10 Uncomplicated urinary tract infections 250 mg every 12 hours 7 to 10 Uncomplicated gonorrhea 1,000 mg single dose Early Lyme disease 500 mg every 12 hours 20 Pediatric Patients younger than 13 years (who can swallow tablets whole) Acute bacterial otitis media 250 mg every 12 hours 10 Acute bacterial maxillary sinusitis 250 mg every 12 hours 10 2.1 Important Administration Instructions Cefuroxime axetil tablets and cefuroxime axetil for oral suspension are not bioequivalent and are therefore not substitutable on a milligram-per-milligram basis [see Clinical Pharmacology (12.3)] . Administer cefuroxime axetil tablets as described in the appropriate dosage guidelines [see Dosage and Administration (2.2) ]. Administer cefuroxime axetil tablets with or without food. Pediatric patients (aged 13 years and older) who cannot swallow the cefuroxime axetil tablets whole should receive cefuroxime axetil for oral suspension because the tablet has a strong, persistent bitter taste when crushed [see Dosage and Administration (2.2) ]. 2.2 Dosage for Cefuroxime Axetil Tablets Administer cefuroxime axetil tablets as described in the dosage guidelines table below with or without food. Table 1. Adult Patients and Pediatric Patients Dosage Guidelines for Cefuroxime Axetil Tablets Infection Dosage Duration (Days) Adults and Adolescents (13 years and older) Pharyngitis/tonsillitis (mild to moderate) 250 mg every 12 hours 10 Acute bacterial maxillary sinusitis (mild to moderate) 250 mg every 12 hours 10 Acute bacterial exacerbations of chronic bronchitis (mild to moderate) 250 mg or 500 mg every 12 hours 10 a Uncomplicated skin and skin-structure infections 250 mg or 500 mg every 12 hours 10 Uncomplicated urinary tract infections 250 mg every 12 hours 7 to 10 Uncomplicated gonorrhea 1,000 mg single dose Early Lyme disease 500 mg every 12 hours 20 Pediatric Patients younger than 13 years (who can swallow tablets whole) b Acute bacterial otitis media 250 mg every 12 hours 10 Acute bacterial maxillary sinusitis 250 mg every 12 hours 10 a The safety and effectiveness of cefuroxime axetil tablets administered for less than 10 days in patients with acute exacerbations of chronic bronchitis have not been established. b When crushed, the tablet has a strong, persistent bitter taste. Therefore, patients who cannot swallow the tablet whole should receive the oral suspension. 2.5 Dosage in Patients with Impaired Renal Function A dosage interval adjustment is required for patients whose creatinine clearance is less than 30 mL/min, as listed in Table 4 below, because cefuroxime is eliminated primarily by the kidney [see Clinical Pharmacology (12.3) ]. Table 4. Dosing in Adults with Renal Impairment Creatinine Clearance (mL/min) Recommended Dosage ≥30 No dosage adjustment 10 to <30 Standard individual dose given every 24 hours <10 (without hemodialysis) Standard individual dose given every 48 hours Hemodialysis A single additional standard dose should be given at the end of each dialysis
