Drug Catalog - Product Detail
AMOXICILLIN & CLAVULANATE POTASSIUM FOR SUSPENSION, USP SUSP 200/28.5MG/5ML 100ML
| NDC | Mfr | Size | Str | Form |
|---|---|---|---|---|
| 00143-9981-01 | HIKMA PHARMACEUTICALS USA | 100 | 200-28.5MG/5ML | SUSPENSION |
PACKAGE FILES


Generic Name
AMOXICILLIN AND CLAVULANATE POTASSIUM
Substance Name
AMOXICILLIN
Product Type
HUMAN PRESCRIPTION DRUG
Route
ORAL
Application Number
ANDA065191
Description
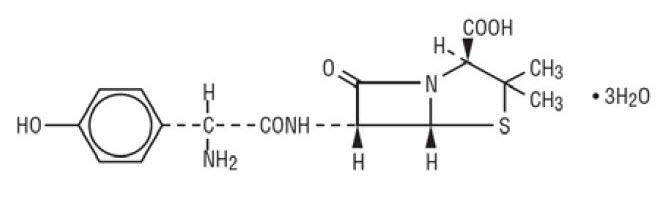
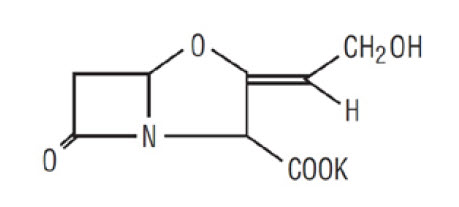
11 DESCRIPTION Amoxicillin and Clavulanate Potassium for Oral Suspension, USP is an oral antibacterial combination consisting of amoxicillin and the beta-lactamase inhibitor, clavulanate potassium (the potassium salt of clavulanic acid). Amoxicillin is an analog of ampicillin, derived from the basic penicillin nucleus, 6-aminopenicillanic acid. The amoxicillin molecular formula is C 16 H 19 N 3 O 5 S•3H 2 O, and the molecular weight is 419.46. Chemically, amoxicillin is ( 2S , 5R , 6R )-6-[( R )-(-)-2-Amino-2-( p -hydroxyphenyl)acetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid trihydrate and may be represented structurally as: Clavulanic acid is produced by the fermentation of Streptomyces clavuligerus . It is a beta-lactam structurally related to the penicillins and possesses the ability to inactivate some beta-lactamases by blocking the active sites of these enzymes. The clavulanate potassium molecular formula is C 8 H 8 KNO 5 , and the molecular weight is 237.25. Chemically, clavulanate potassium is potassium ( Z )( 2R , 5R )-3-(2-hydroxyethylidene)-7-oxo-4-oxa-1-azabicyclo[3.2.0]-heptane-2-carboxylate and may be represented structurally as: Inactive Ingredients: Aspartame, colloidal silicon dioxide, HPMC 2910/ hypromellose 5 cP, silicon dioxide, succinic acid, xanthan gum, golden syrup flavor, orange flavor. Each 5 mL of reconstituted amoxicillin and clavulanate potassium 400 mg/57 mg per 5 mL suspension contains 0.268 mEq of potassium. Each 5 mL of reconstituted amoxicillin and clavulanate potassium 200 mg/28.5 mg per 5 mL suspension contains 0.143 mEq of potassium amoxicillin structural formula clavulanate structural formula
How Supplied
16 HOW SUPPLIED/STORAGE AND HANDLING Amoxicillin and Clavulanate Potassium for Oral Suspension, USP 200 mg/28.5 mg per 5 mL: The dry powder is white to off white with fruity flavor. Each 5 mL of reconstituted creamy suspension contains 200 mg amoxicillin and 28.5 mg clavulanic acid as the potassium salt. NDC 0143-9981-50 .................. 50 mL bottle NDC 0143-9981-75 .................. 75 mL bottle NDC 0143-9981-01 .................. 100 mL bottle Amoxicillin and Clavulanate Potassium for Oral Suspension, USP 400 mg/57 mg per 5 mL: The dry powder is white to off white with fruity flavor. Each 5 ml of reconstituted creamy suspension contains 400 mg amoxicillin and 57 mg clavulanic acid as the potassium salt. NDC 0143-9982-50 .................. 50 mL bottle NDC 0143-9982-75 .................. 75 mL bottle NDC 0143-9982-01 .................. 100 mL bottle Dispense in original container. Store dry powder at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Store reconstituted suspension under refrigeration. Discard unused suspension after 10 days. Keep out of the reach of children.
Indications & Usage
1 INDICATIONS AND USAGE Amoxicillin and Clavulanate Potassium for Oral Suspension is indicated for the treatment of infections in adults and pediatric patients, due to susceptible isolates of the designated bacteria in the conditions listed below: Lower Respiratory Tract Infections -caused by beta‑lactamase‑producing isolates of Haemophilus influenzae and Moraxella catarrhalis . Acute Bacterial Otitis Media -caused by beta‑lactamase‑producing isolates of H. influenzae and M. catarrhalis . Sinusitis -caused by beta‑lactamase‑producing isolates of H. influenzae and M. catarrhalis . Skin and Skin Structure Infections -caused by beta‑lactamase‑producing isolates of Staphylococcus aureus , Escherichia coli , and Klebsiella species. Urinary Tract Infections -caused by beta‑lactamase‑producing isolates of E. coli , Klebsiella species, and Enterobacter species. Limitations of Use When susceptibility test results show susceptibility to amoxicillin, indicating no beta-lactamase production, Amoxicillin and Clavulanate Potassium for Oral Suspension should not be used. Usage To reduce the development of drug‑resistant bacteria and maintain the effectiveness of Amoxicillin and Clavulanate Potassium for Oral Suspension and other antibacterial drugs, Amoxicillin and Clavulanate Potassium for Oral Suspension should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy. Amoxicillin and Clavulanate Potassium for Oral Suspension is a combination of amoxicillin, a penicillin-class antibacterial and clavulanate potassium, a beta‑lactamase inhibitor indicated for treatment of the following infections in adults and pediatric patients: ( 1 ) Lower respiratory tract infections Acute bacterial otitis media Sinusitis Skin and skin structure infections Urinary tract infections Limitations of Use When susceptibility test results show susceptibility to amoxicillin, indicating no beta-lactamase production, Amoxicillin and Clavulanate Potassium for Oral Suspension should not be used. ( 1 ) Usage To reduce the development of drug-resistant bacteria and maintain the effectiveness of Amoxicillin and Clavulanate Potassium for Oral Suspension and other antibacterial drugs, Amoxicillin and Clavulanate Potassium for Oral Suspension should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1 )
Dosage and Administration
2 DOSAGE AND ADMINISTRATION Adults and Pediatric Patients greater than 40 kg: 500 or 875 mg every 12 hours or 250 or 500 mg every 8 hours, based on amoxicillin component. ( 2.2 , 2.3 ) Pediatric patients aged 12 weeks (3 months) and older: 25 to 45 mg/kg/day every 12 hours or 20 to 40 mg/kg/day every 8 hours, up to the adult dose. ( 2.3 ) Neonates and infants less than 12 weeks of age: 30 mg/kg/day divided every 12 hours, based on the amoxicillin component. Use of the 125 mg/5 mL oral suspension is recommended. ( 2.3 ) 2.1 Important Administration Instructions Amoxicillin and clavulanate potassium suspension may be taken without regard to meals; however, absorption of clavulanate potassium is enhanced when amoxicillin and clavulanate potassium suspension is administered at the start of a meal. To minimize the potential for gastrointestinal intolerance, Amoxicillin and clavulanate potassium suspension should be taken at the start of a meal. 2.2 Adult Patients See dosing regimens of Amoxicillin and clavulanate potassium suspension (based on the amoxicillin component) provided in Table 1 below. Table 1. Dosing Regimens of Amoxicillin and clavulanate potassium in Adult Patients TYPE OF INFECTION DOSING REGIMEN OF Amoxicillin and clavulanate potassium Severe infections and infections of the respiratory tract one 875 mg tablet a of Amoxicillin and clavulanate potassium every 12 hours or one 500 mg tablet b,c of Amoxicillin and clavulanate potassium every 8 hours Less severe infections one 500 mg tablet b,c of Amoxicillin and clavulanate potassium every 12 hours or one 250 mg tablet d of Amoxicillin and clavulanate potassium every 8 hours a Adults who have difficulty swallowing may be given the Amoxicillin and clavulanate potassium 200 mg/28.5 mg per 5 mL suspension or the Amoxicillin and clavulanate potassium 400 mg/57 mg per 5 mL suspension may be used in place of the 875 mg/125 mg tablet. b Adults who have difficulty swallowing may be given the Amoxicillin and clavulanate potassium 125 mg/31.25 mg per 5 mL or Amoxicillin and clavulanate potassium 250 mg/62.5 mg per 5 mL suspension in place of the 500 mg/125 mg tablet. c Two Amoxicillin and clavulanate potassium 250 mg/125 mg tablets are NOT substitutable with one 500 mg/125 mg Amoxicillin and clavulanate potassium tablet [see Dosage and Administration ( 2.6 )] . d Amoxicillin and clavulanate potassium 250 mg/125 mg tablet is NOT substitutable with Amoxicillin and clavulanate potassium 250 mg/62.5 mg chewable tablet [see Dosage and Administration ( 2.6 )] . 2.3 Pediatric Patients Based on the amoxicillin component, amoxicillin and clavulanate potassium suspension should be dosed as follows: Neonates and Infants Aged less than 12 weeks (less than 3 months): See dosing regimens of Amoxicillin and clavulanate potassium suspension provided in Table 2 below. Table 2: Dosing Regimens of Amoxicillin and clavulanate potassium suspension in Neonates and Infants Aged Less than 12 Weeks (Less than 3 Months) PATIENT POPULATION DOSING REGIMEN Amoxicillin and clavulanate potassium 125 mg/31.25 mg per 5 mL for oral suspension a Neonates and Infants aged less than 12 weeks (less than 3 months) 30 mg/kg/day every 12 hours a Experience with the Amoxicillin and clavulanate potassium 200 mg/28.5 mg per 5 mL formulation in this age group is limited, and thus, use of the Amoxicillin and Clavulanate Potassium 125 mg/31.25 mg per 5 mL for oral suspension is recommended. Patients Aged 12 weeks (3 months) and Older and Weighing Less than 40 kg: See dosing regimens provided in Table 3 below. The every 12 hour regimen is recommended as it is associated with significantly less diarrhea [see Clinical Studies ( 14.2 )] . The amoxicillin and clavulanate potassium 200 mg/28.5 mg per 5 mL and amoxicillin and clavulanate potassium 400 mg/57 mg per 5 mL for oral suspension and amoxicillin and clavulanate potassium 200 mg/28.5 mg and amoxicillin and clavulanate potassium 400 mg/57 mg chewable tablets contain aspartame and should not be used by phenylketonurics [see Warnings and Precautions ( 5.8 )] . Table 3: Dosing in Patients Aged 12 Weeks (3 Months) and Older and Weighing Less than 40 kg INFECTION DOSING REGIMEN Every 12 hours Every 8 hours Amoxicillin and clavulanate potassium 200 mg/28.5 mg per 5 mL or Amoxicillin and clavulanate potassium 125 mg/31.25 mg per 5 mL Amoxicillin and clavulanate potassium 400 mg/57 mg per 5 mL for oral suspension a or Amoxicillin and clavulanate potassium 250 mg/62.5 mg per 5 mL for oral suspension a Otitis media b , sinusitis, lower respiratory tract infections, and more severe infections 45 mg/kg/day every 12 hours 40 mg/kg/day every 8 hours Less severe infections 25 mg/kg/day every 12 hours 20 mg/kg/day every 8 hours a Each strength of Amoxicillin and clavulanate potassium for oral suspension is available as a chewable tablet for use by older children. b Duration of therapy studied and recommended for acute otitis media is 10 days. Patients Weighing 40 kg or More : Pediatric patients weighing 40 kg or more should be dosed according to adult recommendations. The 250-mg tablet of amoxicillin and clavulanate potassium should NOT be used until the child weighs at least 40 kg, due to the different amoxicillin to clavulanic acid ratios in the 250 mg/125 mg tablet of amoxicillin and clavulanate potassium versus the 250 mg/ 62.5 mg chewable tablet of amoxicillin and clavulanate potassium. 2.4 Patients with Renal Impairment Patients with impaired renal function do not generally require a reduction in dose unless the impairment is severe. Renal impairment patients with a glomerular filtration rate (GFR) of less than 30 mL/min should NOT receive the 875 mg dose (based on the amoxicillin component) of Amoxicillin and clavulanate potassium. See dosing regimens in patients with severe renal impairment provided in Table 4. Table 4. Dosing Regimens of Amoxicillin and clavulanate potassium in Patients with Severe Renal Impairment Patients with Renal Impairment Dosing Regimen GFR 10 mL/min to 30 mL/min 500 mg or 250 mg every 12 hours, depending on the severity of the infection GFR less than 10 mL/min 500 mg or 250 mg every 24 hours, depending on severity of the infection Hemodialysis 500 mg or 250 mg every 24 hours, depending on severity of the infection Administer an additional dose both during and at the end of dialysis 2.5 Directions for Mixing Amoxicillin and Clavulanate Potassium for Oral Suspension Prepare Amoxicillin and clavulanate potassium for oral suspension at time of dispensing as follows: Tap bottle until all powder flows freely. Measure a total (see Table 5 below for total amount of water for reconstitution) OF WATER. Add approximately 2/3 of the water to the powder. Replace cap and shake VIGOROUSLY. Add remaining water. Replace cap and shake VIGOROUSLY. Table 5: Amount of Water for Mixing Amoxicillin and Clavulanate Potassium for Oral Suspension Strength of Amoxicillin and clavulanate potassium for Oral Suspension Bottle Size Amount of Water for Reconstitution Contents of Each Teaspoonful (5 mL) 200 mg/28.5 mg per 5 mL 50 mL 75 mL 100 mL 48 mL 71 mL 95 mL 200 mg of amoxicillin and 28.5 mg of clavulanic acid as the potassium salt 400 mg/57 mg per 5 mL 50 mL 75 mL 100 mL 45 mL 68 mL 90 mL 400 mg of amoxicillin and 57 mg of clavulanic acid as the potassium salt Shake oral suspension well before using. Reconstituted suspension must be stored under refrigeration and discarded after 10 days. Some color change is normal during dosing period. 2.6 Switching between Dosage Forms and between Strengths Amoxicillin and clavulanate potassium 250 mg/125 mg Tablet is NOT Substitutable with Amoxicillin and clavulanate potassium 250 mg/62.5 mg Chewable Tablet The 250 mg/125 mg tablet of Amoxicillin and clavulanate potassium and the 250 mg/62.5 mg chewable tablet of Amoxicillin and clavulanate potassium should NOT be substituted for each other and the 250 mg/125 mg tablet of Amoxicillin and clavulanate potassium should NOT be used in pediatric patients weighing less than 40 kg [see Dosage and Administration ( 2.3 )] . The 250 mg tablet of Amoxicillin and clavulanate potassium and the 250 mg chewable tablet of Amoxicillin and clavulanate potassium do not contain the same amount of clavulanic acid. The 250 mg tablet of Amoxicillin and clavulanate potassium contains 125 mg of clavulanic acid whereas the 250 mg chewable tablet of Amoxicillin and clavulanate potassium contains 62.5 mg of clavulanic acid. Two Amoxicillin and clavulanate potassium 250 mg/125 mg Tablets are NOT Substitutable with One 500 mg/125 mg Amoxicillin and clavulanate potassium Tablet Two 250 mg/125 mg tablets of Amoxicillin and clavulanate potassium should NOT be substituted for one 500 mg/125 mg tablet of Amoxicillin and clavulanate potassium. Since both the 250 mg and 500 mg tablets of Amoxicillin and clavulanate potassium contain the same amount of clavulanic acid (125 mg, as the potassium salt), two 250 mg tablets of Amoxicillin and clavulanate potassium are not equivalent to one 500 mg tablet of Amoxicillin and clavulanate potassium.
